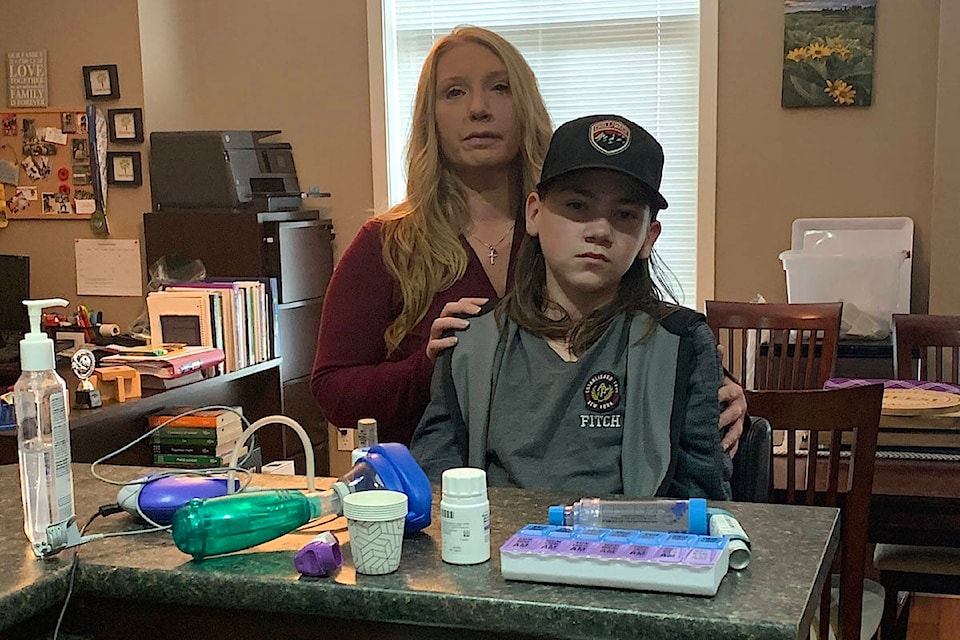
Jocelyn James of Chilliwack launched a national petition to stop federal changes to drug pricing rules that prevent promising new medicines from coming to Canada.
The effort has garnered the support of Chilliwack-Hope MP Mark Strahl, and MP Matt Jeneroux, the Conservative shadow minister for Health.
The Chilliwack mom reached out to MP Strahl to explain the devastating impact that lack of access to new drugs was having on her family.
“After hearing Jocelyn’s concerns, I committed to working with her to raise awareness of this issue through a petition to Parliament. I would encourage anyone concerned about access to new drug treatments to sign her petition,” said MP Strahl in a release.
This week the House of Commons e-petition started by the Chilliwack mom to repeal the drug regulations had earned more than 12,000 signatures.
Steven, the 16-year-old son of Jocelyn James, is someone who loves playing hockey, soccer and being a referee. He also struggles with cystic fibrosis (CF).
“He’s one of the people dramatically affected by this,” James said, estimating that these regulatory changes will impact more than three million Canadians.
Up until the age of 13, Steven was relatively healthy and running track competitively, as well as playing other sports, but saw a decline in the wake of a lung infection a few years ago, his mom told The Progress.
One of the meds currently unavailable in Canada has “revolutionized” treatment for CF patients elsewhere. Becoming known as a “CF wonder drug,” Trikafta is one such medicine.
But the new class of meds, known as “modulators” won’t get approval under the rules, even though they are designed to correct the malfunction in the gene, not just treat the symptoms of CF.
What is at issue is the Patented Medicine Prices Review Board, which sets drug price guidelines. Those PMPRB guidelines are set to change on July 1, 2020 and could result in new drugs being priced below the cost of research, development and production making it impossible to bring them to Canadians.
“New medicines can be a lifeline to those suffering from painful and debilitating illnesses for which conventional medicine has provided little help,” said MP Strahl.
READ MORE: U.S. eyes Canada’s plans to overhaul drug pricing
The proposed price restrictions have already stopped some new treatments from coming to Canada.
The changes being made to the PMPRB in Ottawa could deny Canadians access to life-altering therapies.
“For some it could result in premature death. That’s unacceptable,” Strahl said.
Jeneroux agreed, calling on the federal government to delay the changes.
“Over the past few months, I’ve been hearing from stakeholders and Canadians living with rare diseases that these changes will deter companies from offering their drugs on the Canadian market,” Edmonton Riverbend MP Jeneroux said in a news release. “If the Trudeau government proceeds with their plan, innovative and lifesaving medications could be unavailable for Canadians.”
The petition calls upon the Government of Canada:
“To repeal the changes to the Patented Medicines Regulations that were adopted in August 2019; engage in substantive, transparent and evidence-based consultations with stakeholders on any changes to the Patented Medicine Prices Review Board; and work with patients, clinicians, provincial health systems, researchers and medicine developers to inform the federal price review regime so that the Patented Medicine Prices Review Board does not obstruct access to new medicines and clinical trials.”
To sign the House of Commons petition e-2546, see the details and petition which will close on May 20.
READ MORE: U.S. last year said it would review drug access from Canada
Do you have something to add to this story, or something else we should report on? Email:
jfeinberg@theprogress.com
@CHWKjourno
Like us on Facebook and follow us on Twitter.
Want to support local journalism during the pandemic? Make a donation here.